-
PDF
- Split View
-
Views
-
Cite
Cite
Muhammad Shahzaib, Muhammad Muaz, Muhammad Hasnain Zubair, Masood Ur Rehman Kayani, MiCK: a database of gut microbial genes linked with chemoresistance in cancer patients, Database, Volume 2024, 2024, baae124, https://doi.org/10.1093/database/baae124
Close - Share Icon Share
Abstract
Cancer remains a global health challenge, with significant morbidity and mortality rates. In 2020, cancer caused nearly 10 million deaths, making it the second leading cause of death worldwide. The emergence of chemoresistance has become a major hurdle in successfully treating cancer patients. Recently, human gut microbes have been recognized for their role in modulating drug efficacy through their metabolites, ultimately leading to chemoresistance. The currently available databases are limited to knowledge regarding the interactions between gut microbiome and drugs. However, a database containing the human gut microbial gene sequences, and their effect on the efficacy of chemotherapy for cancer patients has not yet been developed. To address this challenge, we present the Microbial Chemoresistance Knowledgebase (MiCK), a comprehensive database that catalogs microbial gene sequences associated with chemoresistance. MiCK contains 1.6 million sequences of 29 gene types linked to chemoresistance and drug metabolism, curated manually from recent literature and sequence databases. The database can support downstream analysis as it provides a user-friendly web interface for sequence search and download functionalities. MiCK aims to facilitate the understanding and mitigation of chemoresistance in cancers by serving as a valuable resource for researchers.
Database URL: https://microbialchemreskb.com/
Introduction
Cancer is a significant global health concern, ranking among the leading causes of morbidity and mortality worldwide. According to the WHO, cancer accounted for nearly 10 million deaths in 2020 and was ranked as the second leading cause of global deaths. The GLOBOCAN report highlights that there were approximately 19.3 million new cancer cases and 10 million cancer-related deaths in 2020 [1]. Factors contributing to the incidence of cancer include genetic predisposition, lifestyle factors such as diet, obesity, and alcohol consumption, as well as environmental exposures and infections. Multiple treatment strategies are available for cancer, ranging from surgical interventions to chemotherapy, radiation therapy, immunotherapy, and targeted therapies. The approach to treatment often depends on the type and stage of cancer, as well as the patient’s overall health and preferences [2].
Major drugs used to treat cancer include 5-Flurouracil (5-FU), Capecitabine, Gemcitabine, Irinotecan, and Oxaliplatin. 5-FU is a commonly used antimetabolite drug for cancer that causes the inhibition of thymidylate synthase, an enzyme essential for DNA synthesis and repair. Capecitabine, a prodrug of 5-FU, is converted to 5-FU in tumor tissues by the enzyme thymidine phosphorylase [3]. Furthermore, it has replaced 5-FU due to its improved pharmacokinetics and tolerability [4]. Gemcitabine is a nucleoside analog that inhibits DNA synthesis by interfering with the activity of the ribonucleotide reductase enzyme [5]. Irinotecan, a DNA topoisomerase inhibitor, binds to topoisomerase which causes the inhibition of DNA replication and transcription through its active metabolite i.e. SN38 [3]. Oxaliplatin works by forming DNA adducts inhibiting DNA replication, transcription, DNA damage, and apoptosis of cancer cells [3].
Chemoresistance is a major challenge in the successful treatment of cancer patients. The human gut microbiome has recently been attributed to the emergence of chemoresistance in cancer patients [6]. Gut microbial species including Escherichia coli, Bifidobacterium longum, Citrobacter, and Enterococcus faecalis metabolize 5-FU, the most common drug used in treating cancer patients, and alter its efficacy [7]. Another prominent example includes the increased efflux of oxaliplatin by gut microbes through the glucuronidation mechanism by the β-glucuronidase enzyme. This enzyme inactivates SN38 (an activated form of irinotecan) by converting it to SN38G [8]. Fusobacterium nucleatum, another common gut microbial species, promotes chemoresistance by the modulation of autophagy [9]. Lactobacillus and Bifidobacterium have been found to have higher abundance in the progressive disease group compared to the partial response group in cases administered with folinic acid, 5-FU, and irinotecan [10].
Several databases have been designed, considering the importance of the gut microbiome and its interaction with drugs used in chemotherapy. These include PharmacoMicrobiomics which contains 131 records about the effect of drugs on gut microbiome [6]. Microbe-Drug Association Database contains interactions of 1388 drugs, and 180 microbes curated from drug databases and relevant literature [9]. MagMD is also a comprehensive database for metabolic actions of the gut microbiome on drugs, covering 32 678 interactions between 2146 microbes, 36 enzymes, and 219 substrates [11]. To the best of our knowledge, no database currently exists that provides dedicated access to microbial gene sequences potentially involved in chemoresistance, which can support downstream analysis for therapeutic interventions. To address this challenge, we present the Microbial Chemoresistance Knowledgebase (MiCK), a comprehensive database cataloging microbial gene sequences associated with chemoresistance in CRC. MiCK contains 1.6 million sequences of 29 gene types linked to chemoresistance and drug metabolism, curated from recent literature and databases. The database architecture, built using MySQL, supports efficient data search, retrieval, and download functionalities through its user-friendly web interface. MiCK is designed to facilitate the understanding and mitigation of chemoresistance in cancer treatment, serving as an invaluable resource for researchers.
Materials and methods
Identification of gut microbial chemoresistance genes
For the identification of gut microbial genes, potentially associated with chemoresistance, we first performed a literature search in PubMed [8] and Google Scholar [7] using the following keywords: “chemoresistance,” “cancer,” and “gut microbiome.” 16S rRNA-based studies were excluded. Through manual curation, the search results were restricted to only the publications that reported or discussed microbial genes or products that could potentially result in chemoresistance. In addition, enzymes involved in the metabolism of commonly used therapeutics for cancer were searched in the KEGG [12] and MagMD databases [11].
After collecting information on genes and enzymes, we retrieved their sequences from the UniProtKB, and NCBI Gene databases [13]. Full gene name was used for searching sequences in the databases, from the search results, only bacterial sequences were downloaded for downstream analysis. Through this, we identified 29 gene types that could potentially confer chemoresistance. The genes targeting gemcitabine included cytidine deaminase, thymidine kinases (TK), deoxycytidine kinase (dck), histone deacetylase (HDAC), and ribonucleotide reductase (RNR). Cytidine deaminase, produced by Gammaproteobacteria, is known to inactivate Gemcitabine [14]. TK are involved in the salvage pathway capable of synthesizing deoxythymidine by phosphorylation of thymidine thus helping in DNA synthesis, whereas knocking TK2 by small interfering RNA sensitizes the cell to gemcitabine chemotherapy, thus confirming their role in chemoresistance [15]. dck is reported to be involved in limiting the cytotoxic activity of gemcitabine [16]. HDAC6 is reported to be upregulated in colorectal cancer patients and thus plays a role in poor prognosis and survival [17]. RNR consists of two subunits M1 and M2. M1 subunit has been identified in large amounts in microarray profiles of in vivo gemcitabine resistance model, which depicts its role in chemoresistance [18].
The target genes for oxaliplatin include butyrate, cysteine desulfurase (NFS1), metallothionein (MT), glutathione (GLU), and glutathione S-transferase (GST). Butyrate, a short-chain fatty acid produced by gut microbes, has been reported to play a dual role. Its high concentration in oxaliplatin-resistant cell lines can be considered evidence of its role in chemoresistance [19]. NFS1 weakens the sensitivity of cancer cells to oxaliplatin [20], whereas MT are small cystine-enriched molecules with four isoforms, MT1 to MT4. Thiol groups of MT are involved in chemoresistance to oxaliplatin, a drug used to treat CRC through drug detoxification, which prevents cells from apoptosis and accumulation of oxaliplatin [21]. GLU conjugates with oxaliplatin, and this conjugate serves as the substrate for detoxification enzymes [22], whereas GST is a detoxification enzyme for oxaliplatin [23].
Target genes of 5-FU included dihydropyrimidine dehydrogenase (DHP), dutpase, nucleoside diphosphate kinase (NME2), thymidylate synthase (TS), toll-like receptors (TLRs), UMP/CMP kinase, and thymidine phosphorylase. DHP causes catabolism of 5-FU [24], whereas the expression of Dutpase is linked to susceptibility of cancer cells to chemotherapeutics like 5-FU [25]. Knockdown of the NME2 gene increases sensitivity to 5-FU in CRC cell lines [26], whereas 5-FU inhibits the TS, which stops DNA replication and apoptosis of tumor cells. One of the key indicators of chemoresistance to 5-FU is an increase in the concentration of TS in tumor cells [22]. TLR is the main receptor in the pathway that can mitigate the bacterial production of pro-inflammatory cytokines that help in tumorigenesis and can lead to chemoresistance in cancer cells [27]. An active TLR-4-MyD88 signaling pathway could pose a risk for cancer development and serve as a promising target for creating biomodulators to combat chemoresistance [28]. UMP/CMP kinase attenuates the functioning of 5-FU and leads to chemoresistance against it [29]. Thymidine phosphorylase is involved in the activation of 5-FU, but it is also involved in tumor progression through angiogenesis and by avoiding apoptosis, which depicts its role in chemoresistance against 5-FU-based chemotherapy of cancer [30].
Target genes for irinotecan included UDP-glucuronosyltransferases (UGTs), β-glucuronidase (BGU), topoisomerase inhibitors (DTI), and Hydroxyglutarate. UGTs impair the biological activity by glucuronidation and enhancing the water solubility of the drugs [31]. UMP/CMP kinase is an enzyme involved in the phosphorylation of 5-FU, but miR-130-b which is a key epigenetic regulator of BGU, an enzyme produced mostly by four gut bacterial phyla, namely Bacteroidetes, Firmicutes, Verrucomicrobia, and Proteobacteria. BGU induces toxicity in response to irinotecan thus limiting the dose and affecting the efficacy of irinotecan [32]. Most of the chemotherapeutics are topoisomerase-active substances but the DTIs are involved in the chemoresistance against these drugs. DTIs include ATP-binding cassette transporters (ABC), GSH, and HDAC6 [33]. Hydroxyglutarate has been shown to inhibit α-ketoglutarate-dependent dioxygenases, including histone demethylases, which leads to epigenetic changes that can promote tumor progression and chemotherapeutic resistance [34].
The gut microbiome can influence the functionality of drugs by secreting several enzymes [27]. Therefore, to enhance the pool of genes that can potentially result in chemoresistance, we searched for pathways of drug metabolism in the KEGG Pathways [12] and explored the MagMD database that provided the information about interaction of microbial enzymes and drugs [11]. We then retrieved the sequences of all the enzymes from UniProtKB and NCBI gene databases. β-ureidopropionase, phosphoribosyl transferase, uridine phosphorylase, uracil phosphoribosyl transferase, carboxyl esterase, uridine kinase, and deoxycytidine kinase were the enzymes involved in the metabolism of cancer drugs and were used in our database. The list of chemoresistance genes (CRGs) is provided in Table 1.
Chemoresistance genes, target drugs, and the potential effects of the genes
| CRGs . | Drug . | Effect . | Reference . |
|---|---|---|---|
| Butyrate | Oxaliplatin | Chemoresistance | [19] |
| Cytidine deaminase | Gemcitabine | Chemoresistance | [14] |
| Cystine desulfurase | Oxaliplatin | Decreased sensitivity/ Chemoresistance | [20] |
| Dihydropyridine dehydrogenase | Capecitabine/5-FU | Catabolism | [24] |
| Glutathione | Oxaliplatin | Chemoresistance | [35] |
| Glutathione s transferase | Oxaliplatin | Detoxification enzyme | [23] |
| UDP-glucuronosyltransferases | Irinotecan | Chemoresistance | [31] |
| Nucleoside diphosphate kinase | Capecitabine | Chemoresistance | [26] |
| Ribonucleotide reductase | Gemcitabine | Chemoresistance | [18] |
| Thymidine kinases | Gemcitabine | Chemoresistance | [15] |
| Thymidylate synthase | 5-FU | Drug target | [22] |
| UMP/CMP kinase | Capecitabine | Chemoresistance | [29] |
| β-glucuronidase | Irinotecan | Reduced efficacy | [32] |
| ATP-binding cassette transporters | Oxaliplatin | Efflux of drugs | [33] |
| Metallothionein | Oxaliplatin | Chemoresistance | [21] |
| Hydroxyglutarate | Gemcitabine | Chemoresistance | [34] |
| DNA topoisomerase inhibitors | Irinotecan | Chemoresistance | [33] |
| Carboxyl esterase | Capecitabine | Substrate metabolism | [11] |
| dUTPase | Capecitabine | Chemoresistance | [36] |
| Histone deacetylase | Gemcitabine | Chemoresistance | [17] |
| Purine nucleoside phosphorylase | 5-FU | Chemoresistance | [26] |
| Thymidine phosphorylase | Capecitabine | Chemoresistance | [30] |
| Toll like receptors | 5-FU | Chemoresistance | [28] |
| β-ureidopropionase | Capecitabine | Substrate metabolism | [11] |
| Phosphoribosyl transferase | 5-FU | Substrate metabolism | [11] |
| Uridine phosphorylase | Capecitabine | Substrate metabolism | [11] |
| Uracil phosphoribosyl transferase | Capecitabine | Substrate metabolism | [11] |
| Uridine kinase | 5-FU | Substrate metabolism | [11] |
| Deoxycytidine kinase | Gemcitabine | Substrate metabolism | [11] |
| CRGs . | Drug . | Effect . | Reference . |
|---|---|---|---|
| Butyrate | Oxaliplatin | Chemoresistance | [19] |
| Cytidine deaminase | Gemcitabine | Chemoresistance | [14] |
| Cystine desulfurase | Oxaliplatin | Decreased sensitivity/ Chemoresistance | [20] |
| Dihydropyridine dehydrogenase | Capecitabine/5-FU | Catabolism | [24] |
| Glutathione | Oxaliplatin | Chemoresistance | [35] |
| Glutathione s transferase | Oxaliplatin | Detoxification enzyme | [23] |
| UDP-glucuronosyltransferases | Irinotecan | Chemoresistance | [31] |
| Nucleoside diphosphate kinase | Capecitabine | Chemoresistance | [26] |
| Ribonucleotide reductase | Gemcitabine | Chemoresistance | [18] |
| Thymidine kinases | Gemcitabine | Chemoresistance | [15] |
| Thymidylate synthase | 5-FU | Drug target | [22] |
| UMP/CMP kinase | Capecitabine | Chemoresistance | [29] |
| β-glucuronidase | Irinotecan | Reduced efficacy | [32] |
| ATP-binding cassette transporters | Oxaliplatin | Efflux of drugs | [33] |
| Metallothionein | Oxaliplatin | Chemoresistance | [21] |
| Hydroxyglutarate | Gemcitabine | Chemoresistance | [34] |
| DNA topoisomerase inhibitors | Irinotecan | Chemoresistance | [33] |
| Carboxyl esterase | Capecitabine | Substrate metabolism | [11] |
| dUTPase | Capecitabine | Chemoresistance | [36] |
| Histone deacetylase | Gemcitabine | Chemoresistance | [17] |
| Purine nucleoside phosphorylase | 5-FU | Chemoresistance | [26] |
| Thymidine phosphorylase | Capecitabine | Chemoresistance | [30] |
| Toll like receptors | 5-FU | Chemoresistance | [28] |
| β-ureidopropionase | Capecitabine | Substrate metabolism | [11] |
| Phosphoribosyl transferase | 5-FU | Substrate metabolism | [11] |
| Uridine phosphorylase | Capecitabine | Substrate metabolism | [11] |
| Uracil phosphoribosyl transferase | Capecitabine | Substrate metabolism | [11] |
| Uridine kinase | 5-FU | Substrate metabolism | [11] |
| Deoxycytidine kinase | Gemcitabine | Substrate metabolism | [11] |
Chemoresistance genes, target drugs, and the potential effects of the genes
| CRGs . | Drug . | Effect . | Reference . |
|---|---|---|---|
| Butyrate | Oxaliplatin | Chemoresistance | [19] |
| Cytidine deaminase | Gemcitabine | Chemoresistance | [14] |
| Cystine desulfurase | Oxaliplatin | Decreased sensitivity/ Chemoresistance | [20] |
| Dihydropyridine dehydrogenase | Capecitabine/5-FU | Catabolism | [24] |
| Glutathione | Oxaliplatin | Chemoresistance | [35] |
| Glutathione s transferase | Oxaliplatin | Detoxification enzyme | [23] |
| UDP-glucuronosyltransferases | Irinotecan | Chemoresistance | [31] |
| Nucleoside diphosphate kinase | Capecitabine | Chemoresistance | [26] |
| Ribonucleotide reductase | Gemcitabine | Chemoresistance | [18] |
| Thymidine kinases | Gemcitabine | Chemoresistance | [15] |
| Thymidylate synthase | 5-FU | Drug target | [22] |
| UMP/CMP kinase | Capecitabine | Chemoresistance | [29] |
| β-glucuronidase | Irinotecan | Reduced efficacy | [32] |
| ATP-binding cassette transporters | Oxaliplatin | Efflux of drugs | [33] |
| Metallothionein | Oxaliplatin | Chemoresistance | [21] |
| Hydroxyglutarate | Gemcitabine | Chemoresistance | [34] |
| DNA topoisomerase inhibitors | Irinotecan | Chemoresistance | [33] |
| Carboxyl esterase | Capecitabine | Substrate metabolism | [11] |
| dUTPase | Capecitabine | Chemoresistance | [36] |
| Histone deacetylase | Gemcitabine | Chemoresistance | [17] |
| Purine nucleoside phosphorylase | 5-FU | Chemoresistance | [26] |
| Thymidine phosphorylase | Capecitabine | Chemoresistance | [30] |
| Toll like receptors | 5-FU | Chemoresistance | [28] |
| β-ureidopropionase | Capecitabine | Substrate metabolism | [11] |
| Phosphoribosyl transferase | 5-FU | Substrate metabolism | [11] |
| Uridine phosphorylase | Capecitabine | Substrate metabolism | [11] |
| Uracil phosphoribosyl transferase | Capecitabine | Substrate metabolism | [11] |
| Uridine kinase | 5-FU | Substrate metabolism | [11] |
| Deoxycytidine kinase | Gemcitabine | Substrate metabolism | [11] |
| CRGs . | Drug . | Effect . | Reference . |
|---|---|---|---|
| Butyrate | Oxaliplatin | Chemoresistance | [19] |
| Cytidine deaminase | Gemcitabine | Chemoresistance | [14] |
| Cystine desulfurase | Oxaliplatin | Decreased sensitivity/ Chemoresistance | [20] |
| Dihydropyridine dehydrogenase | Capecitabine/5-FU | Catabolism | [24] |
| Glutathione | Oxaliplatin | Chemoresistance | [35] |
| Glutathione s transferase | Oxaliplatin | Detoxification enzyme | [23] |
| UDP-glucuronosyltransferases | Irinotecan | Chemoresistance | [31] |
| Nucleoside diphosphate kinase | Capecitabine | Chemoresistance | [26] |
| Ribonucleotide reductase | Gemcitabine | Chemoresistance | [18] |
| Thymidine kinases | Gemcitabine | Chemoresistance | [15] |
| Thymidylate synthase | 5-FU | Drug target | [22] |
| UMP/CMP kinase | Capecitabine | Chemoresistance | [29] |
| β-glucuronidase | Irinotecan | Reduced efficacy | [32] |
| ATP-binding cassette transporters | Oxaliplatin | Efflux of drugs | [33] |
| Metallothionein | Oxaliplatin | Chemoresistance | [21] |
| Hydroxyglutarate | Gemcitabine | Chemoresistance | [34] |
| DNA topoisomerase inhibitors | Irinotecan | Chemoresistance | [33] |
| Carboxyl esterase | Capecitabine | Substrate metabolism | [11] |
| dUTPase | Capecitabine | Chemoresistance | [36] |
| Histone deacetylase | Gemcitabine | Chemoresistance | [17] |
| Purine nucleoside phosphorylase | 5-FU | Chemoresistance | [26] |
| Thymidine phosphorylase | Capecitabine | Chemoresistance | [30] |
| Toll like receptors | 5-FU | Chemoresistance | [28] |
| β-ureidopropionase | Capecitabine | Substrate metabolism | [11] |
| Phosphoribosyl transferase | 5-FU | Substrate metabolism | [11] |
| Uridine phosphorylase | Capecitabine | Substrate metabolism | [11] |
| Uracil phosphoribosyl transferase | Capecitabine | Substrate metabolism | [11] |
| Uridine kinase | 5-FU | Substrate metabolism | [11] |
| Deoxycytidine kinase | Gemcitabine | Substrate metabolism | [11] |
Non-redundant CRG catalogue and database construction
MMseqs2 (version 13.45111) was used to cluster the CRGs [37]. MMseqs2 was used for indexing the sequence database (generated from the concatenation of all sequences), followed by clustering using linclust. Clustering was performed using multiple identity thresholds (i.e. 50%, 75%, 80%, 90%, and 100%) with query coverage of 80%.
The database was created using MySQL (version 8.0.2, https://www.mysql.com/) and consists of five columns: “Accession_number,” “Gene_type,” “Drug,” “Uniport_Accession,” and “Effects.” The accession number serves as the primary key of the database, the second column contains gene type information, the third column provides the target drug for the gene type, the fourth column holds the full gene type names, the fifth column contains the original accession numbers for retrieving sequences from UniProtKB, and the last column holds the information of effects on the drugs. The framework of the database construction is illustrated in Fig. 1.
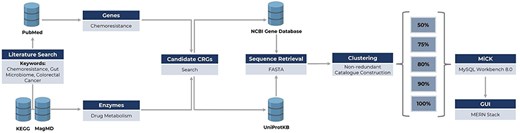
Framework of MiCK development, including (i) literature search for the identification of genes/enzymes linked to chemoresistance, (ii) retrieval of respective gene and enzyme sequences from NCBI Gene and UniProtKB databases, and (iii) sequence clustering for constructing MiCK using MySQL Workbench.
Graphical user interface design
The MiCK interface was developed using the MERN stack, integrating MongoDB as the database, Express.js for the backend, React.js for the front end, and Node.js as the server environment. MongoDB stores data in a flexible, JSON-like format, while Express.js establishes API endpoints for data retrieval and management, ensuring secure communication between the user and the database. React.js builds a responsive interface using reusable components and state management. Node.js handles server-side operations, processing concurrent requests efficiently. The platform’s components are connected through RESTful architecture, with React sending HTTP requests to the Express backend, which interacts with MongoDB to maintain a user-friendly experience.
MiCK statistics
MiCK hosts ∼1.6 million sequences that encompass 29 CRGs, obtained through a literature search and an extensive review of drug-metabolizing enzymes. Among these, GLU is the most frequent gene type included in MiCK with ∼0.6 million representative gene sequences, followed by PRT (∼0.25 million gene sequences) (Fig. 2a). MiCK contains ∼0.6 million genes that target oxaliplatin, followed by gemcitabine for which ∼0.4 million target gene sequences are present in MiCK (Fig. 2b). The effects column comprises chemoresistance, chemosensitivity, and substrate metabolism. Chemoresistance is the most frequent effect with almost 1.3 million repetitions.
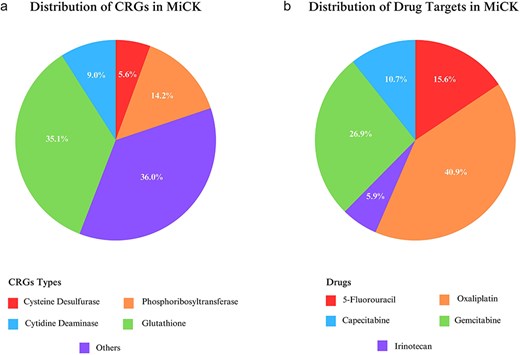
Statistics of distribution of chemoresistance genes (a) and their target drugs (b) included in MiCK.
Database web interface
MiCK offers a user-friendly interface with six navigation bars, i.e. Home, Statistics, Search, Downloads, Team, and About.
Home
The home page welcomes MiCK users to the website and also provides a basic description of cancer and problems associated with chemoresistance, and the necessity to create MiCK. From this page, the users can navigate to various other pages of the website, which include Statistics, Search, Downloads, Teams, and About pages.
Statistics
The statistics page of the MiCK features interactive pie charts that display statistics for chemoresistance genes and drug targets in MiCK. The users can click on the categories, and they will be brought to the search page showing results corresponding to the clicked category. This feature enhances user experience and makes data exploration more efficient.
Search
The search page comprises a selection and search boxes. Through the selection box, the user can select the search criteria, whereas the search box allows keyword input. Users can search through accession numbers, drug names, gene names, and effects. The resultant table displays information on gene type, its drug target, potential effect on the drug, and the accession number of sequences.
Downloads
The downloads page offers the utility to download the CRG catalog, which includes the sequencing files as well as the metadata. Currently, users can download the CRG catalog clustered at 50%, 75%, 80%, 90%, and 100% similarity cutoffs. Metadata includes information about the CRGs, corresponding drugs, and effects on the drug. The graphical user interface of MiCK is shown in Fig. 3.
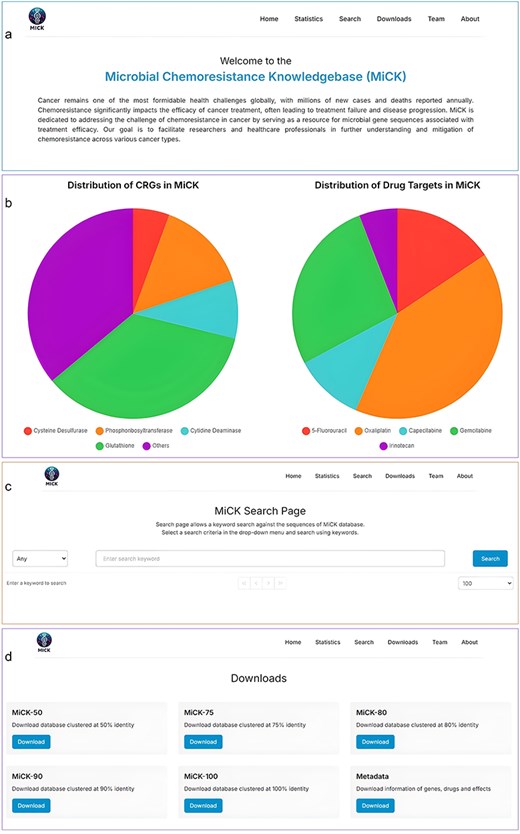
Major components of the MiCK graphical user interface: (a) The Home page, (b) The Statistics page, (c) The Search page, and (d) The Downloads page.
Team
The Team page provides information and contact details for the Principal Investigator, author, co-author, and database developer. This section ensures transparency and facilitates direct communication with the key contributors to the MiCK project.
About
The About page provides help for the users by answering some of the most frequently asked questions that a potential user can have in mind. In addition, the contact information of the database administrator is also provided for further queries.
Acknowledgements
The authors are highly thankful to the members of the Metagenomics Discovery Lab at the SINES, NUST, Islamabad, Pakistan, for useful discussion and feedback on our manuscript.
Conflict of interest
None declared.
Funding
This research was funded by the Graduate Research Support Fund to M.S. (Student Registration No.: 402305) by the National University of Sciences & Technology (NUST), Islamabad, Pakistan.
Data availability
The publicly retrieved gene sequences and relevant details can be downloaded from Zenodo using doi: https://doi.org/10.5281/zenodo.14190584.



